Test Engineer
Role details
Job location
Tech stack
Job description
As a Test Engineer, reporting to the Chief Technology Officer, you'll provide prompt and professional engineering support to Xstrahl therapy and research cabinet systems worldwide. In order to achieve this, your responsibilities will include:
- Attending meetings with the design team to understand the product details (for example, intended use, requirements, software design, and hardware design).
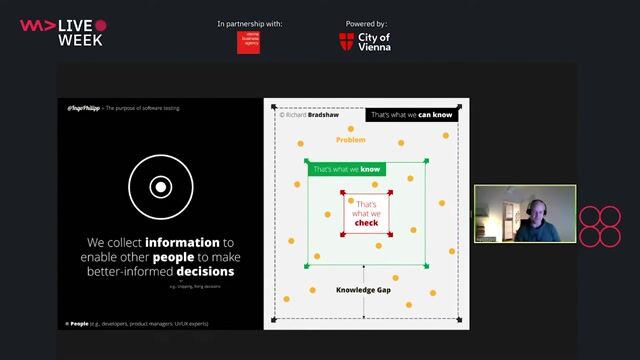
- Developing test strategies.
- Writing test plans and creating test cases, including usability, for the products (software and hardware).
- Analysing opportunities and implementing automated testing.
- Reporting product quality issues and working with the team in resolving the issues.
- Ensuring all tests and procedures meet the Company's Quality Management System and Regulatory standards.
- Preparing test reports for regulatory submission.
Requirements
Do you have experience in Quality management?, Do you have a Bachelor's degree?, An in-depth knowledge of the product development process will be fundamental in this role, as a Test Engineer you'll desire innovative ways to test the product and you'll work with a realisation that you are the last line of defence before the product is released to the market. This will naturally require an individual who has a good eye for details and the ability to think "differently". In addition, you'll also need:
- Bachelor's Degree in Electrical Engineering is preferred
- Other Engineering disciplines with experience or expertise in testing electromechanical systems will be considered.
- Minimum of 2 years' experience in testing software and hardware products.
- Working knowledge of Medical Products Quality Management System.
- Good communication skills and ability to work seamlessly with cross-functional teams.
- Collaborative approach towards problem solving.
- Experience in Medical/Life Sciences radiation products (desirable).
- Experience in Agile Methodology (desirable)
- Experience in creating documentation/report for regulatory compliance (FDA, CE etc) (desirable).
Benefits & conditions
- Competitive salary
- Generous annual bonus scheme
- Life Assurance
- Access to an award winning Employee Assistance Program with 24/7 support
- Opportunity to learn and grow with a personal development plan