Senior Engineer Testing SW/HW
Role details
Job location
Tech stack
Job description
We are seeking a Senior HW/SW Test Engineer to lead and execute verification and validation activities for safety-critical medical devices. In this role, you will be responsible for defining test strategies, developing robust test solutions, and ensuring compliance with global medical device regulations and quality standards. You will work closely with R&D, systems engineering, quality, and regulatory teams throughout the product lifecycle., Test Strategy & Planning
- Define and maintain verification and validation (V&V) strategies for complex HW/SW medical systems
- Derive test requirements from system, software, and hardware specifications
- Ensure full requirements traceability (e.g., per IEC 62304 and ISO 13485)
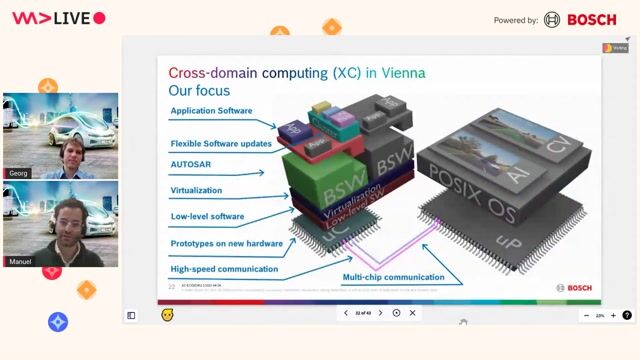
Hardware & Software Testing
- Design, implement, and execute test cases for embedded hardware, firmware, and software
- Perform integration, system, regression, and acceptance testing
- Develop and maintain automated test frameworks (where applicable)
- Specify and build test setups, fixtures, and simulators for HW/SW interaction
Regulatory & Quality Compliance
- Ensure testing activities comply with:
- IEC 62304 (Medical device software)
- ISO 13485 (Quality management systems)
- ISO 14971 (Risk management)
- IEC 60601 (Electrical safety & EMC), as applicable
- Support design reviews, audits, and regulatory submissions (FDA, MDR, etc.)
- Contribute to risk analysis, hazard mitigations, and verification of risk controls
Defect Management & Documentation
- Identify, analyze, document, and track defects through resolution
- Create and maintain test protocols, reports, and technical documentation suitable for regulatory review
- Ensure accurate and timely reporting of test results and quality metrics
Technical Leadership
- Act as a technical mentor for junior test engineers
- Drive continuous improvement in test processes, tools, and methodologies
- Collaborate cross-functionally to resolve complex system-level issues
Requirements
Do you have experience in Test automation?, Do you have a Master's degree?, This role is ideal for an experienced engineer who combines hands-on testing expertise with a strong understanding of medical device standards, risk management, and design controls., * Bachelor's or Master's degree in Electrical Engineering, Computer Engineering, Biomedical Engineering, or a related field
- 5+ years of experience in HW/SW testing, preferably in medical devices or other regulated industries (e.g., aerospace, automotive)
- Strong experience with:
- Embedded systems testing
- Hardware-software integration
- Test case design and execution
- Solid understanding of medical device regulations and standards (IEC 62304, ISO 13485, ISO 14971)
- Experience with requirements and test management tools (e.g., DOORS, Polarion, Jira, TestRail)
- Ability to analyze complex system behavior and troubleshoot across HW and SW boundaries
- Fluent in English (written and spoken)
Preferred / Nice to Have
- Experience with test automation (e.g., Python, LabVIEW, C/C++, CI pipelines)
- Hands-on experience with electronic measurement equipment (oscilloscopes, logic analyzers, power analyzers)
- Knowledge of FDA Design Controls and EU MDR requirements
- Experience in safety-critical or life-sustaining medical devices
- ISTQB or equivalent testing certification
- Experience working in cross-site or global development teams
The anticipated base pay range for this position is 80,400 EUR to 127,765 EUR.
Required Skills:
Preferred Skills: Agility Jumps, Analytical Reasoning, Coaching, Collaborating, Critical Thinking, Model-Based Systems Engineering (MBSE), Product Design, Report Writing, Requirements Analysis, Research and Development, SAP Product Lifecycle Management, Scripting Languages, Software Architectures, Software Development Management, Software Engineering Practices, Software Quality Assurance (SQA), Systems Development Lifecycle (SDLC), Technologically Savvy