Biopharmaceutics in silico modeler
Role details
Job location
Tech stack
Job description
Johnson & Johnson Innovative Medicines seeks to bring innovative and effective treatments in four therapeutic areas of high unmet medical need (oncology, immunology, neuroscience, cardiopulmonary). The Janssen campus in Beerse, close to Antwerp, (Belgium) has a unique ecosystem covering the complete drug development life cycle, with all capabilities from basic science to market access on one campus.
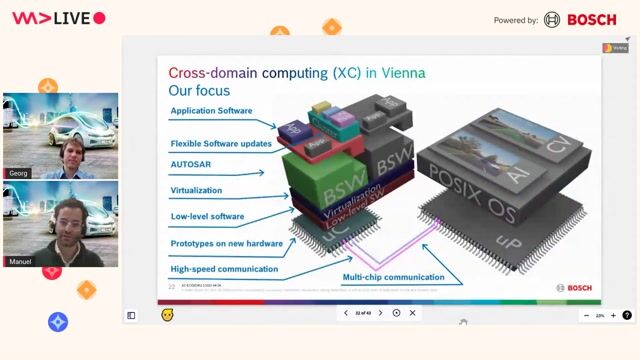
The biopharmaceutics group is part of Pharmaceutical & Material Sciences in Pharmaceutical Product Development & Supply. The group supports the drug product design process for both early and late development projects (for synthetic molecules) by characterizing and optimizing the link between the physicochemical properties of the active ingredient, drug product and its in vivo behavior.
You will be responsible for:
- Building and applying computational models to:
- characterize the interface between the drug product and the physiological target (for multiple delivery routes)
- predict drug absorption and disposition based upon physiological, physicochemical and in vitro release input data (eg via Physiology Based Biopharmaceutical and/or Pharmacokinetics modeling)
- Supporting the selection of the appropriate patient-centric drug delivery system and route of administration to assure optimal delivery of the active ingredient at the site of action
- Actively cooperating with external partners including academic and private research organizations to stay at the forefront of emerging scientific, industrial, and regulatory standards in the field of biopharmaceutics
- Building and demonstrating strong partnerships with key stakeholders including Discovery Sciences, Preclinical DMPK, Non-Clinical Safety, Clinical Pharmacology, Regulatory Affairs, API Development, Drug Product Development and Analytical Development
Requirements
- Master or Doctorate (Ph.D.) in Pharmaceutical Sciences, Chemistry, Biology, Engineering or related science preferably with relevant industrial experience (> 2 years) in biopharmaceutics and computational modeling (drug absorption and disposition).
- Knowledge in (some of) of the following fields is recommended: physico-chemistry, drug absorption/disposition, formulation development/design, pharmacokinetics
- Scientific specialist via a single contributor role, with excellent interpersonal, communication and collaborative skills, while demonstrating a high degree of personal accountability.
Required Skills:
Preferred Skills: Coaching, Collaborating, Critical Thinking, Drug Discovery Development, Goal Attainment, Market Research, Organizing, Pharmaceutical Sciences, Pharmaceutical Trends, Product Development, Product Strategies, Report Writing, Research Documents, Research Proposals, Scientific Research, Standard Scientific Processes and Procedures, Technologically Savvy