IT Engineer
Role details
Job location
Tech stack
Job description
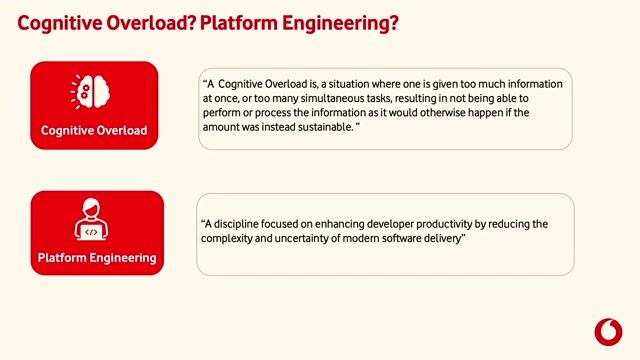
In this role, you will join a dedicated project team supporting an Obsolescence Program, responsible for the structured and controlled retirement of legacy systems and equipment. You will coordinate and oversee the decommissioning of pharmaceutical laboratory and manufacturing IT assets, ensuring full compliance with internal policies, regulatory requirements, and established IT governance processes.
This position requires close collaboration with stakeholders across IT, Quality, Engineering, Manufacturing, and Laboratory functions, as well as strong ownership and accountability for project execution, from initial planning through successful completion., * Handling of Incident, Service Request, Problem ticket and Change Request.
- Preparing compliance report
- Directing validation activities
- Analyzing and documenting test results
- Making adjustment or improvement to equipment, processes, applications, or software
- Interpreting customer requirements
- Developing validation schedules
- Conducting training and overseeing the work of validation technicians
- Keeping up to date on industry standard and regulations
- User Access Management
- License Management
- Backup of the Standalone Bench top systems on regular basis
- SOP and WI updates
- Backup & Restoration for the Lab systems on demand
- Participate in CAPA & Deviation Events
- Support Compliance/Data Integrity related activities
- Interface issues coordination with cross functional team
- Decommissioning and up-gradation of the Legacy system. etc.
Requirements
Do you have experience in Manufacturing?, We are seeking a French-speaking IT Industrial Engineer for an on-site position in Bulle, Switzerland. The successful candidate will bring strong IT expertise combined with hands-on experience in manufacturing and/or laboratory environments, preferably within the pharmaceutical industry.
The ideal candidate is a highly motivated and proactive professional, demonstrating innovation, autonomy, and excellent interpersonal, communication, and problem-solving skills. A strong ability to collaborate effectively across multidisciplinary teams and operate within a regulated environment is essential., * Strong knowledge of pharmaceutical laboratory and manufacturing systems
- Proven experience in support, covering incident, problem, and change management activities
- Experience collaborating with project team members to ensure timely delivery of project activities
- Skilled in preparation and maintenance of support documentation
- Demonstrated analytical and problem-solving skills, with the ability to operate efficiently under pressure
- Highly organized, goal-oriented, trustworthy, and reliable, with strong communication skills in a virtual/online environment
- Experience collaborating with project team members to ensure timely delivery of project activities
- Fluent in both French and English (written and spoken).