Clinical Data Validation Engineer
Role details
Job location
Tech stack
Job description
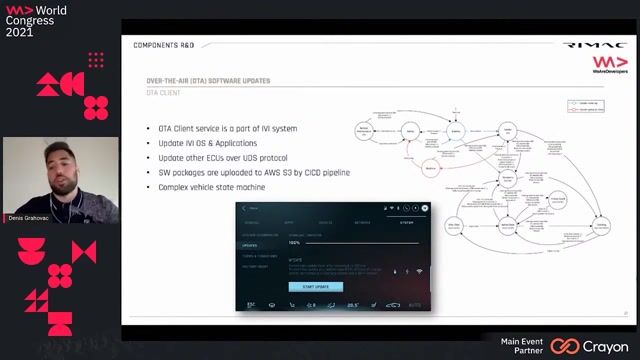
asset for all deliverables. Develop a library of reusable checks working closely with data engineers and config specialists. Collaborate with cross functional teams in creating business rules and automated check library across TAU. Complies with applicable SOPs and work practices. Serves as a technical resource for creating data analytics to help with data validation and cleaning. Supports knowledge development of others on reporting tools that support the accuracy and integrity of study data. Provide programming expertise for data cleaning to efficiently ensure high quality data. Enhances available reporting tools/macros/libraries or creates new ones to support the and review of clinical trial data and trial status information for cross-functional use and provides input on data management reporting standards as required. Creates and tests listings for data review. Ensure operational excellence in collaboration with partners and colleagues for application of standards and develop data, looking for? Even if the perfect role isn't available right now, we're always excited to connect with talented individuals. Register to our Talent Community to receive updates on roles that align with your career goals. Responsibilities The Clinical Data Validation Engineer will create data cleaning reporting solutions and visualizations to support clinical trial activities. They will also develop reusable check libraries and provide programming expertise to ensure the accuracy and integrity of clinical trial data.
Requirements
Clinical data validation, Data cleaning, Reporting tools, SQL, SAS, R, Python, CQL, Veeva CDB, JReview, Elluminate, CDISC, SDTM, Clinical trial terminology, Data visualization, Metadata Rave, cleaning reports in support of the data review plan, in collaboration with the Clinical Data Management and medical reviewers. Your Profile: Bachelor's degree in computer science, statistics, biostatistics, mathematics, biology or other health related field or equivalent experience that provides the skills and knowledge necessary to perform the job. Knowledge of drug development process. Understanding regulatory requirements and relevant data standards; CDISC and SDTM knowledge and experience are preferable. Minimum 2 years' experience in programming listings and/or visualizations with any of the following Veeva CDB, JReview and Elluminate Hands-on experience with report development in Elluminate preferred. Experience with one of these languages: CQL, SQL, SAS, R, Python Knowledge of clinical database systems (Metadata Rave, Veeva, InForm). Proficiency in the use of Microsoft Office Suite of tools (Outlook, Word, Excel, etc.) Strong working knowledge of clinical trial terminology
Benefits & conditions
Must be able to work in a fast-paced environment. Ability to work independently, take initiative and complete tasks to deadlines. What ICON can offer you: Our success depends on the quality of our people. That's why we've made it a priority to build a diverse culture that rewards high performance and nurtures talent. In addition to your competitive salary, ICON offers a range of additional benefits. Our benefits are designed to be competitive within each country and are focused on well-being and work life balance opportunities for you and your family. Our benefits examples include: Various annual leave entitlements A range of health insurance offerings to suit you and your family's needs. Competitive retirement planning offerings to maximize savings and plan with confidence for the years ahead. Global Employee Assistance Programme, TELUS Health, offering 24-hour access to a global network of over 80,000 independent specialised professionals who are there to